so we picked the last one - "copper plating".
This was more of a paragraph in the chapter text than an actual exercise.. it just had a bare description of the copper plating process and the chemical reactions involved.
No wonder our teacher was skeptical about it and told us that we would probably flunk if we didn't do one of the more "reliable" exercises(the graphite one and the stupid electric bell)
Anyway dday arrived and after a really long hour during which each kid showed off their ridiculously identical and lame graphite conduction and electric bell projects it was our turn.
unfortunately we had only managed to secure one of the key ingredients(copper sulphate) that very day.. so we hadn't even tested the rig yet! but we didn't tell the teacher that. we confidently set up the rig on the floor next to her desk(i guess she was a bit concerned about the "toxicity" of the shimmering blue compound.

i dumped a key that i wanted plated, and a volunteer from the audience(we sort of felt like magicians doing a show) gave us some small metal thing(think it was a earing or something) and we flipped the switch.
in a few minutes the metal objects turned a flakey orange-yellow... APPLAUSE!
So heres how it works-
you need
1) atleast 3volts(2 AA batteries) for it to work well. will work with 1.5v(1 AA battery) too, but will be really slow. Will probably work really well with more power.
2) a carbon electrode. we got ours from an old battery. its the inner black colored core.
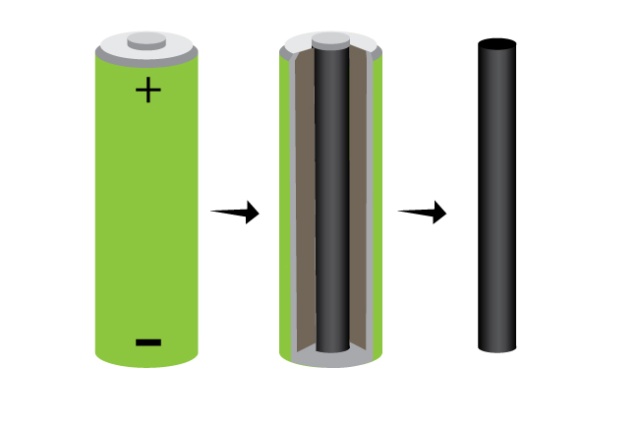
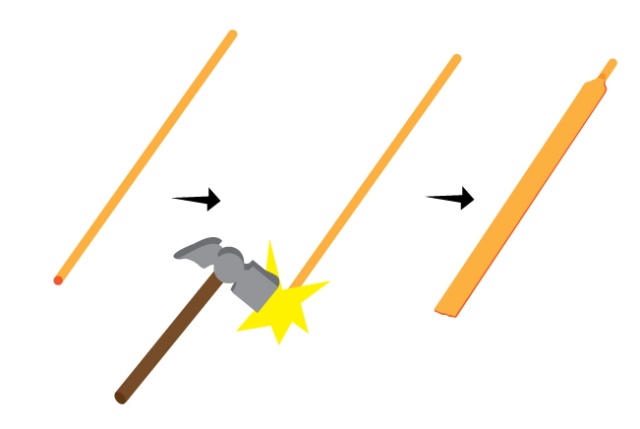
3) a copper electrode. we hammered a bit of copper wire till it was flat. a bit of copper tube might work too.
4) wires
5) a jar/beaker
6) water
7) copper sulphate. we ended up getting some from the chemistry lab.
step1 - connect the copper electrode to the positive side of the battery, and dip it in the copper sulphate sollution(copper sulphate crystals dissolved in water).
step2 - connect the carbon electrode to the negetive end of the battery and dip it in the sollution too.
step3 - drop the metal object you want to plate into the sollution.
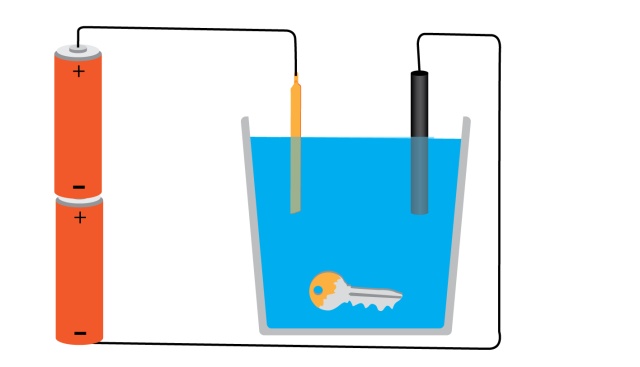
thats it!
UPDATE:added it to instructables.com (see below)
simple Copper plating - More DIY How To Projects
Another really cool chemistry DIY i found today was this water powered calculator by angelo10 at Instructables.com -
Water Powered Calculator - More DIY How To Projects
read the whole DIY thing here
And few more of my favourites:
DIY Electric Bike Conversion - More DIY How To Projects
My home made solar cell step by step - More DIY How To Projects
and a DIY solar pannel using broken solar panel bits that you can order from siliconsolar.com
DIY Solar Panel - More DIY How To Projects
No comments:
Post a Comment